Reshaping the cell membrane with molecular pegs
Researchers at EMBL Hamburg and CSSB's Protein Characterisation facility reveal how peg-like proteins clasp and reshape the cell membrane.
The cell membrane is a highly dynamic structure that constantly changes its shape. The ability to reshape the membrane – which scientists call ‘remodelling’ – is critical for the fundamental biological process of endocytosis. Endocytosis allows cells to take in external materials, such as nutrients, by surrounding them with an area of cell membrane.

The main endocytic machinery relies on coordination in space and time of numerous proteins that connect the membrane to the cytoskeleton, a network of protein filaments and tubules that gives structure to the cells. An essential piece of this 3D puzzle is to understand how adaptor proteins, which anchor the membrane, manage to grip it with enough strength to reshape it.
In a recent study, the García Alai team and collaborators used single particle cryo-electron microscopy to solve a part of this puzzle by determining the molecular structure of the protein anchors. They saw that the structure of these anchors resembles clothes pegs. In further experiments, they found that these pegs combine into larger multi-peg clusters, which clasp the membrane tightly and enable it to be remodelled during endocytosis.
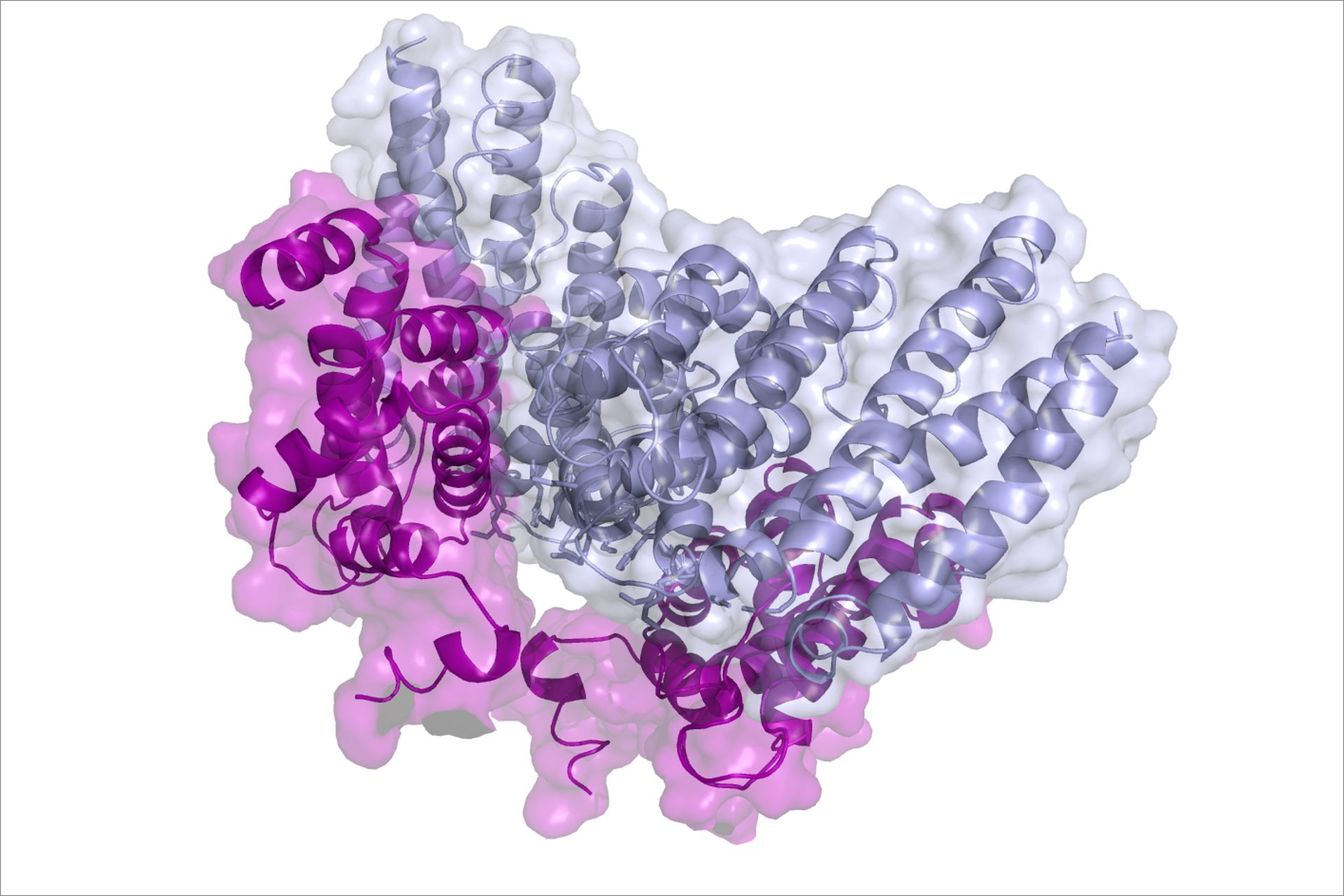
“These findings finally enabled us to understand how adaptor proteins, which are not an integral part of the membrane, are able to reshape it,” says María García Alai, the leading scientist in this study. Javier Lizarrondo, the first author adds: “We also saw that these anchors assemble very fast which is crucial for the cell to respond quickly to the changing environment”.
Source:
Lizarrondo, J., Klebl, D.P., Niebling, S. et al. Structure of the endocytic adaptor complex reveals the basis for efficient membrane anchoring during clathrin-mediated endocytosis. Nat Commun 12, 2889 (2021). https://doi.org/10.1038/s41467-021-23151-7
Original News Story:
https://www.embl.org/news/science/molecular-pegs/



